Isotopic analysis of groundwater tracers
Isodetect offers the analysis of all tracers for groundwater dating. We carry out the measurements in the BioCity Leipzig, in the environmental laboratories of our Helmholtz partners UFZ or HMGU or with cooperating partner laboratories that are specialized in certain tracers.
Recently, some tracers (14C, 18O, 2H, 85Kr) can be analyzed using new, fluorescence-based methods that require significantly smaller amounts of sample. This makes sampling easier, but can lead to significantly longer processing times.
Brief protocols for sampling and analysis of the main groundwater tracers
Tritium
Sampling and storage until analysis is done in 1 L glass or plastic containers. To measure tritium in H2O, the samples are electrolytically enriched and, after adding a scintillation cocktail, analyzed spectrometrically (liquid scintillation counting, LSC). Each sample is measured up to ten times for at least 100 minutes each and then the mean value is determined. Additional standards and blank samples are used to evaluate the data. The inaccuracy of the entire measurement method is ± 0.35 TU (standard deviation) for low tritium levels (<7 TU) and approx. 10% for higher tritium levels >7 TU. At 1 TU, a sample has an activity of 0.119 Bq/L.
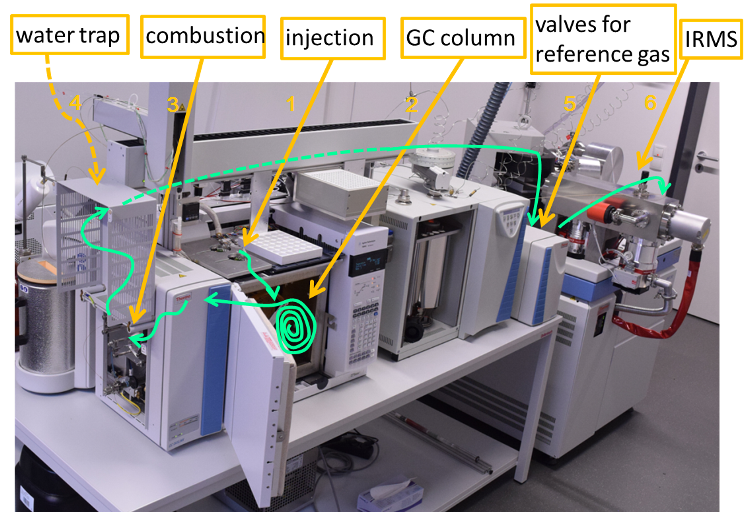
oxygen-18 and deuterium
Samples were taken and stored until analysis in 20 ml glass or plastic containers. The determination of oxygen (18O) or deuterium (2H) in groundwater is carried out on a laser absorption device (e.g. PICARRO L2130-iδD/δ18O Ultra High-Precision). Here, an energy loss of a laser beam caused by the respective isotopes is determined. The measured values of the samples (10 individual measurements) are calibrated with international standards (SLAP, SMOW, GISP) and any drift or memory effects are corrected. The general measurement error is ±0.1‰ for 18O/16O or ± 0.5‰ (standard deviation) based on the VSMOV standard.
The method is limited to samples with no oil contamination and normal or low salinity (no seawater). For such samples we use a different measurement method (EA/Pyr-IRMS).
Carbon-14
Carbon-14 and carbon-13 are determined using accelerator mass spectrometry (AMS; Scharf et al. 2007). The mean measurement inaccuracies are 0.26 ‰ for pm 14C (percent modern) and 0.65 ‰ for δ13C.
This determination takes about 3 months.
Helium-3, Helium-4 and Neon
To determine helium isotopes and neon, approx. 40 mL groundwater is filled into a copper tube which is sealed with screw clamps. The dissolved gases are extracted from the water samples and transferred to a glass ampoule. After separation of helium and neon in a multi-stage cryogenic system, 4He, 20Ne and 22Ne are determined in a quadrupole mass spectrometer and 3He and 4He in a sector field mass spectrometer (Syltenfuss et al. 2009, The Bremen Mass Spectrometric Facility for the measurement of helium isotopes , neon, and tritium in water, Isotopes in Environmental and Health Studies 45(2), 1-13). Defined amounts of air are used for calibration.
CFCs and sulfur hexafluoride
To determine CFC and SF6, 1 L of groundwater is filled into ground glass bottles with the exclusion of air and free of gas bubbles. The bottles are transported in groundwater-filled metal containers. SF6 and CFC are determined using gas chromatography (ECD) according to Oster, Sonntag, Münnich (1996).
Krypton-85
To determine krypton-85, several cubic meters of water are degassed on site with a special device and collected in a gas tank. After drying the gas sample with a molecular sieve, the sample is concentrated and enriched to 85Kr by cryogenic distillation (passing through an activated carbon trap cooled with liquid nitrogen) and subsequent gas-chromatic purification. Subsequently, the 85Kr isotopes of approx. 10 µL krypton are recorded by means of laser excitation in a magneto-optical trap (so-called ATTA analysis; atom trap trace analysis). The detailed analysis protocol is discussed by Tu et al. (2014).
These determinations may take up to 6 months.