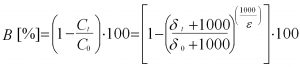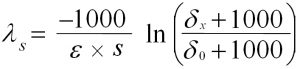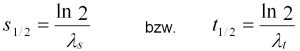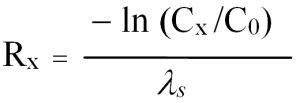Basics
Stable isotopes are light and heavy atoms of the same element.


On earth, stable isotopes occur in an extraordinarily constant ratio, which is typical for each element. The percentage of heavy isotopes in chemical molecules (e.g. H2O or pollutants) as well as in the elements is usually very low (e.g. 13C = 1.11%; Deuterium D=3H= 0.015%). However, in a reactive compound, various chemical, physical and biological processes can lead to a minimal (yet easily measurable) shift in the ratio of heavy to light isotopes (the isotopic signature), i. e. isotope fractionation takes place.


Specifying the isotope signature in delta ‰ describes the difference (quotient) between the isotope ratios of a sample and a worldwide reference standard (above). Other scales (below) are the simple quotient (13C/12C) or the percentage (atomic %) of isotope ratios.
Isotopic values: units and measurement principle
Stable isotopes are measured as a ratio between a heavy and a light isotope (e.g. 13C/12C) and are given in the so-called delta notation (δ‰) (see Fig. Left). A reference standard is always measured during the analysis, thus achieving high measurement accuracy and sensitivity.
The reference substance for 13C/12C is the so-called Vienna PeeDee Belemnite (VPDB), a limestone from North America. By definition, it has an isotopic signature of 0 ‰. Its 13C isotope percentage is 1.11% and thus its 13C/12C isotope ratio (Rstd) of 0.01122. A δ-permille corresponds approximately to an isotope percentage of 0.001% (10 ppm). An isotope signature of -1 δ‰ means that the 13C/12C ratio of the measured sample is 0.000011 lower than the reference standard VPDB. Most pollutants have a δ13C value between -30‰ and -20‰.
The measurement accuracy for determining stable isotopes is extremely high.
In component-specific isotope determination (CSIA) from environmental samples, the isotope ratio of individual components is determined from a mixture of compounds. First, the components are separated by gas chromatography, followed by combustion into simple molecules (e.g. CO2), which are then subjected ionization and separation of the heavy and light isotopes in a high-vacuum magnetic field. According to these measurement principles, the devices used are called GC-C-IRMS (gas chromatographic combustion isotope ratio mass spectrometer) or HPLC-C-IRMS (high performance liquid chromatograhy).
The standardized measurement error when determining 13C is <0.5‰, i.e. an isotopic enrichment greater than 0.0005% in the remaining pollutant is a significant change. Since hydrogen is less ionizable, the measurement accuracy for the deuterium content of molecules is only 2-5‰. However, this inaccuracy is often compensated for by a much stronger isotope fractionation during biological pollutant degradation.
The sensitivity of conventional IRMS devices for determining the 13C content of a substance is about 1 nmol C (measuring volume 1 µ< /span>L). For low-molecular pollutants such as BTEX or PCE, this results in a detection limit of around 100 µg ml-1. Heavily contaminated groundwater samples can therefore be examined immediately, while extraction methods are used for lower concentrations of pollutants. Depending on the pollutant, solid-phase sorption/desorption, hydrophobic elution or gas purge (“purge and trap” methods) are used here. These methods can also be used to increase the sensitivity for measuring the isotope fractionation of larger molecules.
Isotopic fractionation: biological degradation of pollutants results in a heavier (i.e. more positive) isotopic signature for most pollutants
The occurrence of heavy isotopes in a molecule (e.g. 13C in a pollutant) means that its cohesion increases minimally. This makes the enzymatic cleavage of the molecules with heavy isotopes somewhat more difficult and they accumulate in the remaining pollutant during degradation. The percentage of 13C in the pollutant can e.g. B. from 1.09% to 1.10%, expressed in δ‰ values, it increases from -19.3‰ to -10.2‰. Depending on the enzyme system, this so-called isotope fractionation is more or less strong. Each enzyme that can split a pollutant leads to a specific, constant enrichment factor (ε) that is independent of the temperature and the growth conditions of the microorganisms.
In particular, the steric location of the heavy isotope within a molecule disposes of the enzymatic cleavage. Only when the isotope is at the reactive site of the molecule targeted by the enzyme can significant isotopic fractionation occur. For example, the probability that a 13C atom will occupy the reactive site in a pollutant decreases with the number of all C atoms in the molecule. The smaller the reacting pollutant molecule, the greater the fractionation effect. For large molecules (>12 carbon atoms), the biological isotope fractionation is therefore usually below the detection limit.
During the synthesis of a substance (e.g. a pollutant), the incorporation of heavy isotopes into the molecules depends on certain kinetic conditions. This can lead to a marginal, still measurable shift in the percentage of isotopes in this substance. Different, e.g. B. geographically or technically induced synthesis conditions therefore produce different isotope signatures of the same substance. On this basis, different sources of contamination can be identified in a contaminated site.
Physical processes such as evaporation (e.g. in the case of deuterated water) or desorption can also cause changes in the isotope content of the substance quantities involved.
Equ. 1: Rayleigh-Equation
Equ. 2: Calculation of percentual degradation
Equ. 3: Calculation of the spatial degradation rate constant
Eq. 4: Calculation of the degradation rate constant over time
Eq. 5: Calculation of half-value distance and time
Eq. 6: Prediction of the distance Rx until pollutant concentration falls below a limit value Cx.
Equations for the quantitative calculation of pollutant degradation from isotopic enrichment
The change in the isotope signature of a pollutant allows the quantification of microbial degradation. The so-called Rayleigh equation (Eq. 1; equations also in US EPA 2008) is used to calculate microbial turn-over.
dt and d0 denote the isotope ratio of a compound at a certain time (t) and at the beginning of the microbial conversion (t = 0). Ct and C0 indicate the concentrations of the compound at the beginning of the degradation reaction (t = 0) and after a certain degradation time (t). The enrichment factor e represents the proportional relationship between the changes in isotopic signature and in concentration. It is specific to i) each pollutant and ii) certain degradation conditions (e.g. aerobic, sulphate-reducing, iron-reducing etc.; temperature). The percentage degradation of pollutants (biodegradation; B [%]) between two measuring points located in a contaminated aquifer in an outflow line is determined analogously to Equation 1 from the change in the isotope ratio and a suitable enrichment factor (e). (Eq. 2). The calculation provides the change in concentration Ct/C0 caused by biological degradation between a measuring point with a high pollutant concentration (GWM0) and one in a downstream measuring point with low pollutant concentration (GWMt). The calculated degradation refers to the surface area that results from the spread as a result of dilution/dispersion effects up to the measuring point GWMt . For the sake of simplicity, the estimation assumes that the enrichment factor is constant and representative of the biodegradation along the groundwater flow path under consideration. Furthermore, a possible influence of additional pollutant sources with different initial isotope signatures is neglected.
Eq. 2 describes the temporal change in the isotope signature (isotope enrichment from d0 to dt) for two measuring points in a contaminant plume. Provided there is a direct hydrogeological connection between a highly contaminated measuring point (usually the source of the pollutant) and a measuring point (x) located in its groundwater outflow at a distance of s, isotope enrichment can occur in a well-defined spatial dimension (from d0 to dx). A transformation of Eq. 2 allows the spatial degradation constant ls</strong to be calculated > (Eq. 3). This first-order parameter (exponential degradation) describes the biological pollutant elimination as decreasing concentration per meter flow distance [m-1].
To determine the degradation constant over time lt[ d-1], the transport time on the considered flow section must be known. It results from the GW flow rate (v), the distance between the measuring points (s) and possibly the substance-specific retardation factor r.
The calculation of the temporal in situ degradation constant of the first order lt [d is analogous to the spatial degradation -1] by changing the isotope ratio and a suitable enrichment factor (Eq. 4).
lt describes the rate of pollutant reduction over time through biodegradation between two measuring points and is thus an important tool to assess the behavior of pollutants in a contaminated aquifer. To assess the degradation kinetics, the estimated degradation constants can be compared with data from literature studies.
The degradation constants can also be interpreted as the biological half-life (t1/2) or half-value distance (s1/2), which indicate the time or the flow range in which the pollutant concentration is halved due to biological degradation (Eq. 5).
The product of the degradation constants ls or lt with the pollutant concentration (Ct) in the downstream measuring point also provides the absolute degradation rates ([μg L-1 m-1] or [μg L-1 y-1] at the measuring point. In contrast to rate constants, they notably change with concentration. They describe the decrease in concentration without taking into account the permanent supply of pollutants from the source of contamination. These degradation rates thus not indicate a theoretical decrease in concentration (per meter flow distance or per year) at the measuring point, since the existing concentration is largely maintained by the inflow from the pollutant source.
Furthermore, the flow distance can be determined that is required until pollutant concentration falls below a certain limit value (Cx) due to biological degradation. This so-called reaction space (R) is calculated with Eq. (6):
Certain environmental conditions cause inaccuracies in the quantification of degradation.
Limitations in the quantitative in situ detection of biological pollutant degradation using stable isotope fractionation result from several factors in addition to analytical limitations. In principle, however, a carbon isotope fractionation >1‰ is qualitatively sufficient evidence of pollutant degradation. For a quantitative assessment, the hydrogeological circumstances of the pollutant release and the groundwater transport should be known.
Parallel physical processes such as sorption and evaporation affect the isotopic content of a substance in addition to biological fractionation. However, in groundwater, these changes are 1-2 orders of magnitude smaller than biological effects. In addition to biological degradation, humic substances (as an adsorption matrix) or low water saturation (as an evaporation space) can cause minor inaccuracies for the quantitative assessment of biodegradation.
Also chemical degradation processes can lead to significant fractionation (e.g. in the chemical reduction of chlorinated ethenes by Fe0, which is used in cleaning groundwater barriers). In this respect, a significant isotope fractionation does not necessarily reflect biological degradation. Its detection can also be a check for chemical degradation.
Different redox conditions (i.e. the availability of electron acceptors such as O2, NO3, SO4, FeIII, HCO3) often activate different enzyme systems in the degrading microorganisms. Under oxic conditions, for example, dioxygenases are frequently activated, which lead to a low isotope fractionation of 13C, while under anoxic conditions mostly strongly fractionating monooxygenases serve to the break down of pollutants. In heterogeneous and oxic soil zones, in which the redox conditions change, the degradation performance of the microorganisms can therefore be relatively independent of specific isotope fractionation factors.
The succession or mixture of different isotopic signatures of the same pollutant complicates the interpretation of an isotopic fractionation. A masking of the biological processes could result, if different sources of a pollutant were successively released during the contamination. This can be the case when a leaking tank has been filled with toluene from different geographical origins (and therefore with different isotope content).
Mixing of two isotopic species of a pollutant results in multiple contamination events when the pollutant is both a contaminant and a degradation product. For instance, in the case of multiplex contamination with chlorinated ethenes (PCE-TCE-DCE-VC), which are simultaneously successively dehalogenated, only qualitative statements on biological degradation are possible for the last three pollutants.